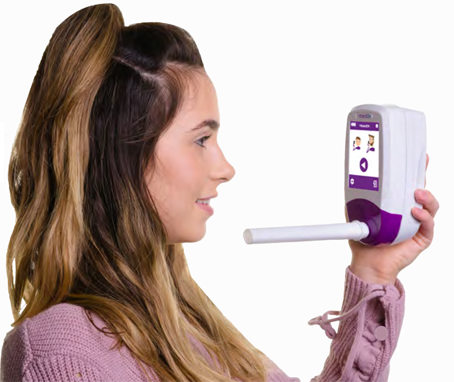
Bedfont’s NObreath® monitor for airway inflammation has been cleared for sale in the USA UK, med-tech company, Bedfont Scientific Ltd., is celebrating a brighter 2022 as the U.S. Food and... read more →
UK, med-tech company, Bedfont Scientific Ltd., is celebrating a brighter 2022 as the U.S. Food and Drug Administration (FDA) cleared its NObreath FeNO monitor for use in monitoring airway inflammation.... read more →
NHS Organisations can apply for funding from the Pathway Transformation Fund (PTF) to obtain a NObreath® used for asthma diagnosis and management. The NObreath® FeNO monitor from Bedfont® Scientific Ltd.... read more →
Funding Opportunities for NHS Organisations NHS Organisations can apply for funding from the Pathway Transformation Fund (PTF) to obtain a NObreath® used for asthma diagnosis and management. NObreath® FeNO Device:... read more →
The Trouble with Diagnosing Asthma Approximately 339 million people across the globe suffer from asthma, and asthma-related deaths affect the lives of three families daily. Of these, up to two-thirds... read more →
Bedfont announces launch of 2nd generation NObreath at ERS 2018, Paris Bedfont Scientific Ltd. launches their new FeNO monitor exclusively at the world’s largest European Respiratory Society Congress. Medical device... read more →
Bedfont Scientific Ltd. is exhibiting its wide range of medical devices at Arab Health 2019, including its new and improved NObreath® FeNO monitor Breath analysis medical device manufacturer, Bedfont, will... read more →
Bedfont’s long-standing UK distributor, Intermedical UK, will be exhibiting their breath analysis medical products at the annual ARTP conference. Intermedical UK, a bistributor for nearly 10 years, will be exhibiting... read more →